Sickle cell disease (SCD) pathophysiology involves uncontrolled inflammation, dysregulated coagulation, and adherence of erythrocytes, leukocytes, and platelets to endothelium, all contributing to disrupted rheology and ultimately vaso-occlusion. Crizanlizumab (Novartis), an FDA-approved humanized monoclonal antibody, binds to p-Selectin and blocks endothelial interactions with blood cells. The phase 2 SUSTAIN trial demonstrated a reduction in the median rate of pain crisis, as well as prolonged time to first and second crisis ( Ataga 2017). Not all participants benefited, however, nor have all treated patients achieved pain reduction in the post-marketing period. Paradoxical infusion-related pain has also been reported ( Kanter 2022). Baseline differences between responders, non-responders, and those suffering infusion-related pain remain uncharacterized. In January 2023, Novartis issued a preliminary update on the ongoing global crizanlizumab phase 3 STAND trial which indicated no statistically significant pain reduction with crizanlizumab therapy compared to placebo. A better understanding of patient suitability for the various disease modifying therapies for SCD would however be beneficial. Our center is a pilot site for a lab conducting an assay measuring flow adhesion of whole blood to P-selectin (FA-WB-Psel) utilizing a protein coated microfluidic chamber by Functional Fluidics. Previously published data identified a steady state reference range for FA-WB-Psel in non-SCD as well as SCD patients ( Pittman 2021). The SMAART Criz study aims to characterize downstream biological effects of crizanlizumab (FA-WB-Psel) and identify biomarkers with the potential to predict treatment response, with the goal of facilitating more individualized prescribing.
This is an interim analysis of an investigator-initiated, prospective, IRB-approved, 18-week longitudinal study at the Children's Hospital of Michigan. Patients desiring crizanlizumab treatment were offered enrollment. Following informed consent, samples were obtained for FA-WB-Psel before and after each crizanlizumab dose. Flow adhesion of whole blood to vascular cell adhesion molecule-1 (FA-WB-VCAM) was assessed as a control substrate. Thromboelastography (TEG) and whole blood platelet aggregometry were performed to evaluate platelet function at baseline, week 6, and week 18. Patient reported response to treatment was used to measure response to therapy.
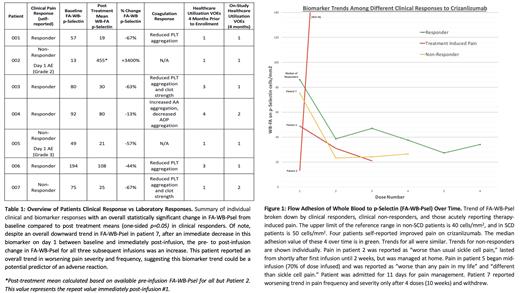
As of August 2023, 7 patients have completed the study with a median age of 20 years (range 16-25 years; 4 female). Genotypes are HbSS (5), HbSC (1), and HbS-G-Philadelphia (1). Four of 7 patients self-reported clinical response to therapy (Table 1). Treatment was discontinued after 1st dose due to treatment-related acute pain in 2 participants, and after the 4th dose due to patient-reported worsening pain trend (Figure 1). Clinical responders had a median baseline FA-WB-Psel adhesion index of 86 cells/mm 2 (n=4, mean 106, range of 56-193). Clinical responders experienced a 65% reduction in FA-WB-Psel from baseline (mean 86) to the mean pre-infusion level calculated using visit 2 and 3 levels (p=0.05, later visits affected by dropout). Non-responders had a median baseline FA-WB-PSel of 49 cells/mm 2 (n=3, mean 46, range 13-75) with widely variable FA-WB-PSel responses (Fig. 1). The two patients with acute treatment-induced pain had the lowest baseline values at 13 and 49 cells/mm 2. No significant trends were observed in adhesion to a control substrate using FA-WB-VCAM. There was an overall decrease in both arachidonic acid (AA) and adenosine diphosphate (ADP) platelet aggregation over time (AA: 23.5 Ohms to 11.3 Ohms, p=0.254; ADP: 23.4 Ohms to 10.6 Ohms, p=0.036).
Early indications from SMAART Criz are that administration of crizanlizumab decreases FA-WB-Psel immediately after infusion with continued decreases over time. Interim analysis suggests elevated baseline FA-WB-Psel may predict individuals with SCD likely to benefit from crizanlizumab, while normal FA-WB-Psel levels at baseline may predict non-response or even adverse reactions. This cohort showed an overall decrease in both AA and ADP aggregation, indicating reduced platelet aggregation following crizanlizumab treatment, a potential downstream biological change contributing to treatment effect (Table 1). Further investigation is warranted. Enrollment and analyses are ongoing.
Disclosures
Chitlur:HRSA/MCHB: Research Funding; BPL Inc: Honoraria; Novo Nordisk: Consultancy, Honoraria; Genzyme Corp: Honoraria; Genentech Inc: Honoraria, Research Funding; Takeda: Honoraria; Children's Foundation: Research Funding; Agios Pharmaceuticals: Honoraria, Research Funding; Novartis Pharmaceuticals: Research Funding. Hines:Functional Fluidics: Current Employment, Current equity holder in private company. Zaidi:Functional Fluidics: Current Employment. Borhan:Functional Fluidics: Current Employment. Glaros:Bausch: Membership on an entity's Board of Directors or advisory committees; Global Blood Therapeutics: Membership on an entity's Board of Directors or advisory committees.